Abstract
Introduction: Acute myeloid leukemia (AML) is the most common type of acute leukemia in adults, accounting for almost 80 percent of the cases. Incidence of AML increases with age and it ranges from 3 to 5 cases per 100,000 persons in the United States. Advances in treatment have led to significant improvements in outcomes for younger patients, while prognosis in the elderly remains poor. There are different sociodemographic and clinical factors that have an impact on survival such as type of leukemia (secondary vs novo), low socioeconomic status, age, gender, health regimen, co-morbidities and performance status
Objective: The aim of the study was to describe the impact of sociodemographic and clinical factors on survival of patients with AML in 11 health institutions from Colombia, from 2009 to June 2021.
Methods: Population based on RENEHOC (online platform) and PETHEMA (Spanish Program for Hematology Treatments). Kaplan-Meier analysis was used to assess overall survival (OS) and Relapse-Free Survival global (RFS) of the different evaluated factors.
Results: A total of 463 patients were included. The median age at diagnosis was 61 years (range, 19-90) and 50.5% were female. According to the FAB classification, 95 (26.6%), 84 (23.6%), and 53 (14.9%) of patients were classified as M2, M0 and M1, respectively. The cytogenetic risk was applied for 227 patients (57%), 135 (59.5%) were intermediate and 78 (34.4%) were high-risk. Secondary AML were 73 (18.2%) and these cases evolved from hematological malignancies in 38 cases (80.8%), the most common were myelodysplastic syndrome (n=16; 34%) and chronic myeloid leukemia (n=7; 50%).
For induction therapy, 232 (59.7%) patients received 7+3 (cytarabine/idarubicin), 47 (11.7%) received Azacitidine (AZA) and 23 (5.7%) received FLUGA (Fludarabine/cytarabine low doses). Complete remission (CR) after induction was achieved in 53% of patients, 12% had partial remission, 20.3% had primary refractory AML. Twelve percent died during induction. The most common consolidation regimen was high dose cytarabine (HiDAC), 143 (35,6%) and 36 (9%) of patients received 1 and 2 cycles respectively.
Seventy-two (51%) patients that achieved a CR relapsed, and 46 (69.7%) received second line therapy. The most common treatment was FLAG-IDA (27%), followed by best supportive care (23.8%). The response rate was 40% (CR:31.1%/PR:8.9%) with 16 (36.6%) patients being refractory to treatment. Five (11.1%) died during salvage therapy.
Thirty-eight (21%) patients had a hematopoietic stem cell transplantation (HCT), 35 (92%) had allogeneic HCT and 3 (8%) autologous HCT, respectively.
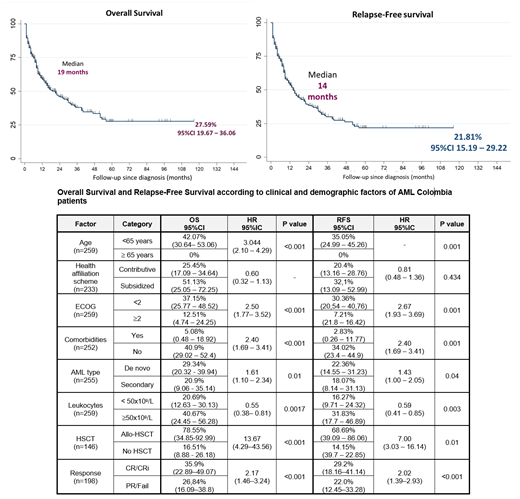
The median and 5-year OS for the whole population was 19 months and 27.6% (95%CI,19.7- 36.0). The median and 5-year RFS was 14 months and 21.8% (95%CI, 15.2 - 29.2), respectively (Figure 1). Sociodemographic and clinical factors such as age, ECOG PS, co-morbidies (Hypertension, diabetes, and chronic heart failure), AML subtype and leukocytosis at diagnosis were prognostic (Table 1).
Conclusion: This is the first multicenter report analyzing real world data from AML patients in Colombia. Results confirm the impact of clinical factors: age, ECOG, secondary LMA on OS and RFS. Challenges includes low alloHSCT rate and low access to complete cytogenetic and molecular classification at diagnosis.
Sossa: Amgen: Research Funding. Abello: Dr Reddy's: Research Funding; Janssen: Honoraria; Amgen: Honoraria. Peña: Amgen: Research Funding. Salazar: Amgen: Research Funding. Sandoval-Sus: SeaGen, Janssen, MassiveBio, TG: Other: Advisory Board; BMS: Other: Advisory Board, Speakers Bureau. Montesinos: Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Agios: Consultancy; Tolero Pharmaceutical: Consultancy; AbbVie: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Glycomimetics: Consultancy; Astellas Pharma, Inc.: Consultancy, Honoraria, Other: Advisory board, Research Funding, Speakers Bureau; Forma Therapeutics: Consultancy; Daiichi Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Incyte: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Karyopharm: Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Pfizer: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Sanofi: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Teva: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Stemline/Menarini: Consultancy.